Cellular Senescence Detection Kit - SPiDER-βGal

Cellular Senescence Detection Kit - SPiDER-βGal
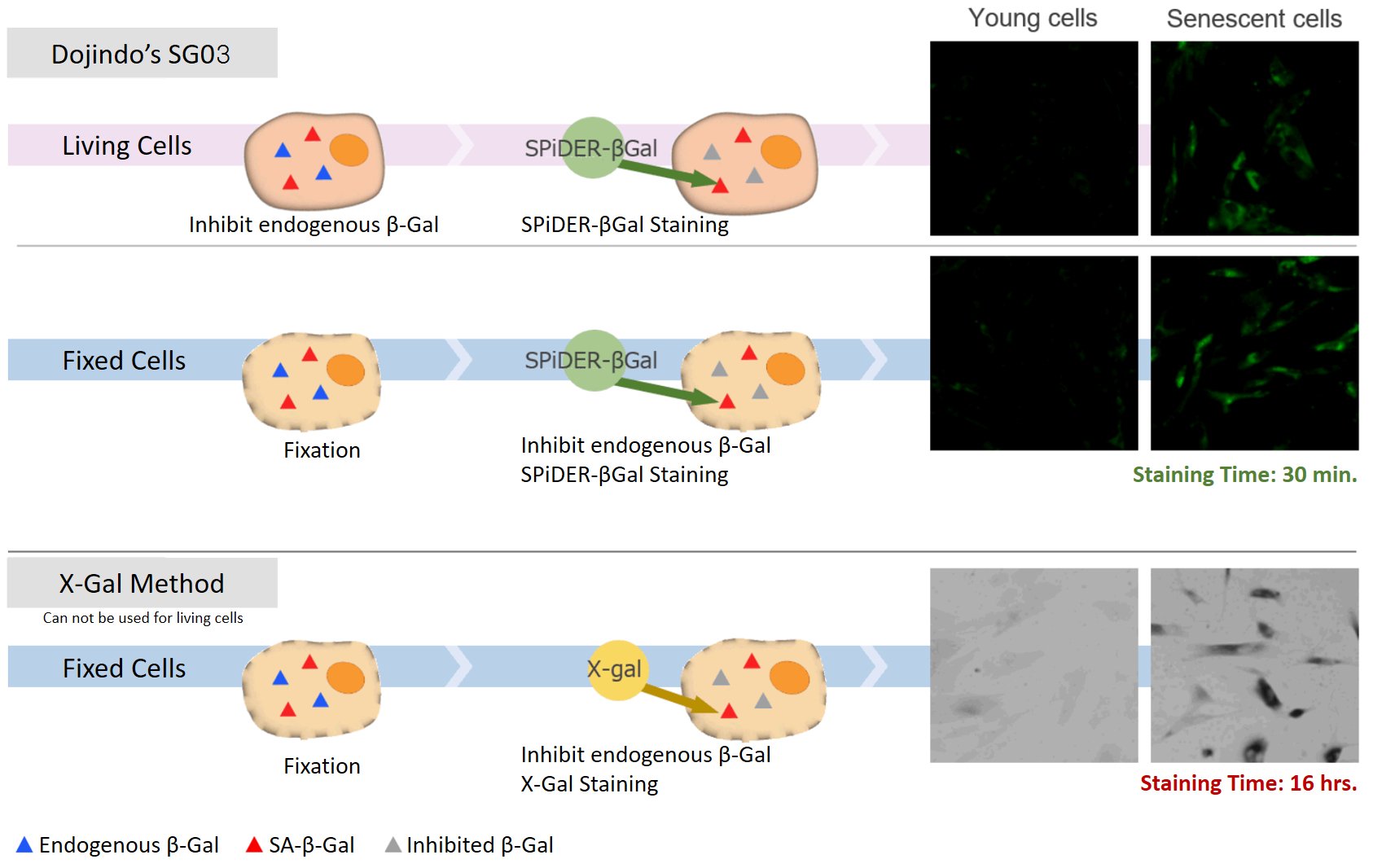
細胞の生存および死をコントロールするために備わった機能として、アポトーシスやネクローシス、オートファジーは、細胞内機能を理解する上で非常に重要です。X-galはSA-β-galを検出するためのよく知られた試薬ですが、1)細胞透過性が低いために固定細胞が必要、2)視覚的測定が難しいために定量能力が低い染色された細胞と染色されていない細胞の違い、3)長時間の染色の必要性といった欠点がありました。
細胞老化検出キット–SPiDER-βGalを使用すると、SA-β-galを高感度で使いやすく検出できます。 SPiDER-βGalは、細胞透過性が高く、細胞内の保持力が高いβ-ガラクトシダーゼを検出するための新しい試薬です。
製品情報
老化細胞検出試薬・キット(傾向顕微鏡、FCMによる高感度解析)

DNA damages of the normal cells are caused by repeated cell division and oxidative stress. Cellular Senescence, a state of irreversible growth arrest, can be triggered in order to prevent DNA-damaged cells from growing. Senescence-associated β-galactosidase (SA-β-gal), which is overexpressed in senescent cells, has been widely used as a marker of cellular senescence. Although X-gal is a well known reagent to detect SA-β-gal, these are following disadvantages: 1) requirement of fixed cells due to the poor cell-permeability, 2) low quantitative capability because of the difficulty of the determination of visual difference between stained cells and not stained cells, 3) requirement of a long time of staining.
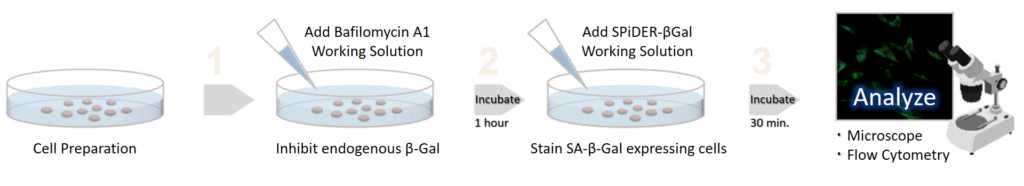
Cellular Senescence Detection Kit – SPiDER-βGal allows to detect SA-β-gal with high sensitivity and ease of use. SPiDER-βGal is a new reagent to detect β-galactosidase which possesses a high cell-permeability and a high retentivity inside cells. SA-β-gal are detected specifically not only in living cells but also fixed cells by using a reagent (Bafilomycin A1) to inhibit endogenous β-galactosidase activity. Therefore, SPiDER-βGal can be applied to quantitative analysis by flow cytometry.
Recent work from Dr. Kim et al. at Mayo Clinic used our Cellular Senescence Detection Kit – SPiDER-βGal to evaluate cellular senescence in endothelial cells. They did staining of SA-βGal in cells in atherosclerotic renal artery stenosis (ARAS) and co-stained the cells with CD31 which is a marker of endothelilal cells. They showed that ARAS + Elamipretide* treatment slightly improved endothelial cell senescence. Unlike commercial available probes used for detection of β-Galactosidase, SPiDER-βGal contained in the kit possesses high intracellular retention. The key feature of this product is that it can be used to co-stain SA-β-Gal and other markers. Our kit is a useful tool for cellular senescence research.
*Elamipretide: mitochondria-targeted peptide
For more information on data, please refer to the publication below:
S. R. Kim, A. Eirin, X. Zhang, A. Lerman and L. O. Lerman, “Mitochondrial Protection Partly Mitigates Kidney Cellular Senescence in Swine Atherosclerotic Renal Artery Stenosis.”, Cell. Physiol. Biochem. ., 2019, 52, 617.
Technical info

Simple Procedure
Difference between X-Gal method and Cellular Senescence Detection Kit – SPiDER-βGal I
Why is Bafilomycin A1 added?
Endogenous β-galactosidase existing in living cells interfere with selective detection of SA-β-Gal. Bafilomycin A1 is an inhibitor of ATPase in lysosome. pH in lysosome is kept neutral by adding Bafilomycin A1. Cellular Senescence Detection Kit - SPiDER-βGal contains Bafilomycin A1 which allows to detect SA-β-Gal selectively. Bafilomycin A1 is utilized for living cell assays only. Bafilomycin A1 is not used in fixed cells because intracellular pH is controlled with the buffer.
Difference between X-Gal method and Cellular Senescence Detection Kit - SPiDER-βGal II
Our kit allows quantification of SA-β-Gal using flow cytometry.

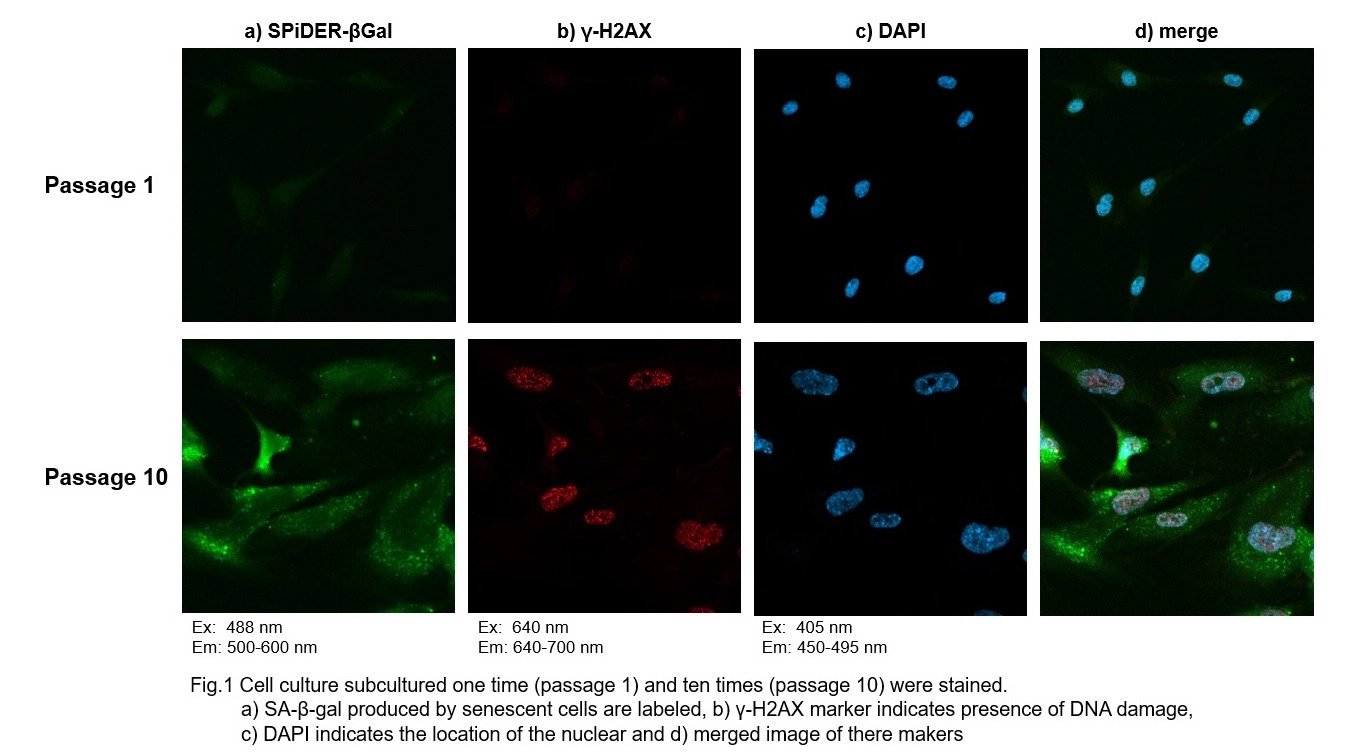
Co-staining of SA- β-gal and DNA Damage marker in WI-38 cells
Procedure:
1. Passage 1 and 10 of WI-38 were used. The procedure was followed as the manual within the kit.
2. Add 4% PFA/PBS to the cells and incubate for 15 minutes at room temperature
3. Wash the cells 3 times with PBS
4. Add 0.1% Triton X-100/PBS to cells and incubate for 30 minutes at room temperature
5. Wash the cells 3 times with PBS
6. Add 1% BSA/PBS to the cells and incubate for 1 hour at the room temperature
7. Add anti- γ-H2AX antibody (rabbit) diluted with 1% BSA/PBS to the cells and incubate at 4℃ overnight
8. Wash the cells 3 times with PBS
9. Add Anti- rabbit secondary antibody (Alexa Fluor 647) diluted with 1% BSA/PBS to the cells and incubate at room temperature for 2 hours
10. Wash cells 3 times with PBS
11. Add 2 μg/ml DAPI (code: D523) diluted with PBS to the cells and incubate for 10 minutes at room temperature
12. Wash cells 3 times with PBS and observe under a confocal microscope
Co-staining of SA- β-gal and DNA Damage marker in fixed WI-38 cells
Preparation of SPiDER-βGal working solution
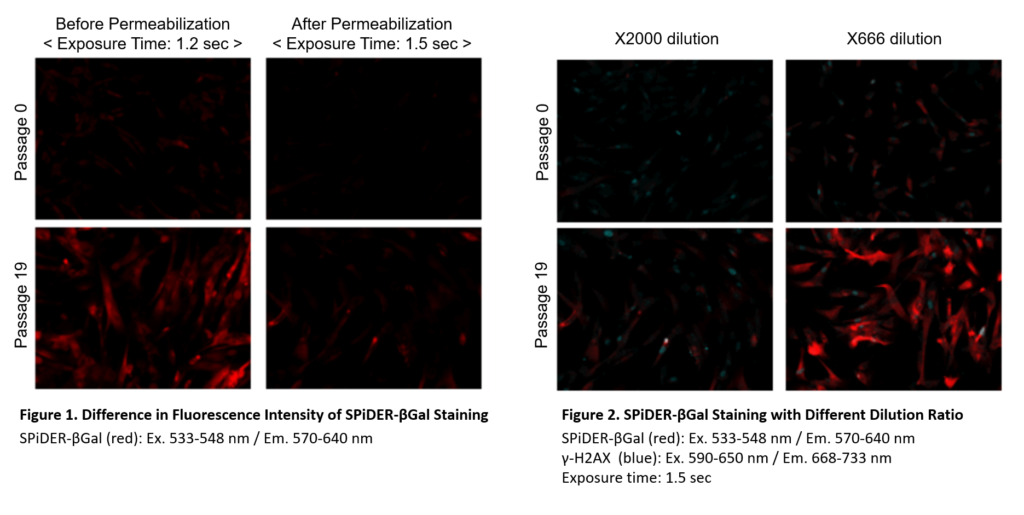
Dilute the SPiDER-βGal DMSO stock solution 2,000 times *1 with McIlvaine buffer (pH 6.0).
*1 Fixation and permeablization could leads to lower sensitivity (Figure 1), if you need higher signals,dilute the SPiDER-βGal DMSO stock solution 500 – 1,000 times with the McIlvaine buffer (Figure 2).
Preparation of McIlvaine buffer (pH 6.0)
Mix 0.1 mol/l citric acid solution (3.7 ml) and 0.2 mol/l sodium phosphate solution (6.3 ml). Confirm the pH is 6.0. If the pH is not 6.0, adjust the pH by adding either citric acid solution or sodium phosphate solution. Dilute this buffer 5 times with ultrapure water.
Staining procedure (35 mm dish)
1. Prepare cells on 35 mm dish for assay and culture the dish at 37℃ overnight in a 5% CO2 incubator.
2. Remove the culture medium. Add 2 ml of 4% paraformaldehyde (PFA) /PBS solution to the cells and incubate at room temperature for 3 minutes *2.
*2 Avoid a longer treatment period, which leads to decrease in SA-β-gal activity.
3. Remove the supernatant, and wash the cells 3 times with 2 ml of PBS.
4. Add 2 ml of SPiDER-βGal working solution and incubate at 37℃ for 30 minutes*3.
*3 We recommend not to use a 5% CO2 incubator for fixed cell experiments. If incubation is done in a 5% CO2 incubator, the pH of the buffer may become acidic. Acidic pH results in higher background from the endogenous β-galactosidase activity and it would be difficult to distinguish between normal cells and senescent cells.
5. After removing the supernatant, wash the cells twice with PBS.
6. Add 0.1% Triton X-100/PBS to cells and incubate for 30 minutes at room temperature.
7. Wash the cells twice with PBS.
8. Add 1% BSA/PBS to the cells and incubate for 1 hour at the room temperature
9. Add anti- γ-H2AX antibody (mouse) diluted with 1% BSA/PBS to the cells and incubate at 4℃ overnight.
10. Wash the cells 3 times with PBS.
11. Add anti- mouse secondary antibody (Cy5) diluted with 1% BSA/PBS to the cells and incubate at room temperature for 1 hour.
12. Wash cells twice with PBS and observe under a fluorescence microscope.
Markers of Senescent Cells

SA-β-gal detection for tissue
In this published article, SA-β-gal was detected using SPiDER-βGal on tissue samples from a diabetic model mouse.
Note: SPiDER-βGal [Code: SG02] is used in this staining.
<Staining Conditions>
After slicing the frozen tissue, it was fixed with 4% paraformaldehyde for 20 minutes at room temperature. Then it was washed with PBS and observed.
For details of the experimental operation and data, refer to Reference 2) below.
SA-β-gal detection for T cells (floating cells)
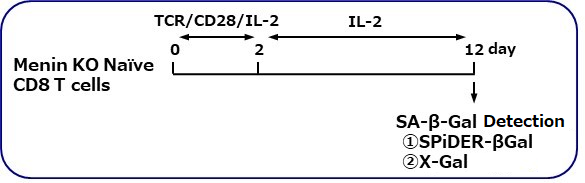
The research group by professor Masakatsu Yamashita at Ehime University Graduate School of Medicine has shown that a protein called Menin controls T cell exhaustion, aging, and maintains normal immune function.
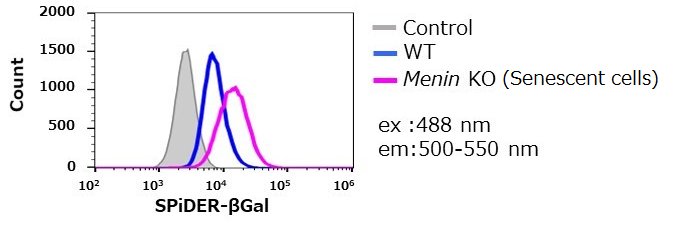
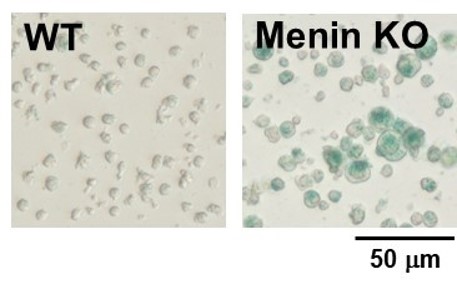
By using this kit, they confirmed that SG03 has the ability to detect induced cell senescence by stimulating TCR (T cell receptor) in the presence of interleukin 2 (IL-II) in naive CD8+ T cells that are knocked out Menin.
Staining Conditions
① SPiDER-βGal method
② X-gal method
*Data was kindly provided by Masakatsu Yamashita, at Ehime University Graduate School of Medicine
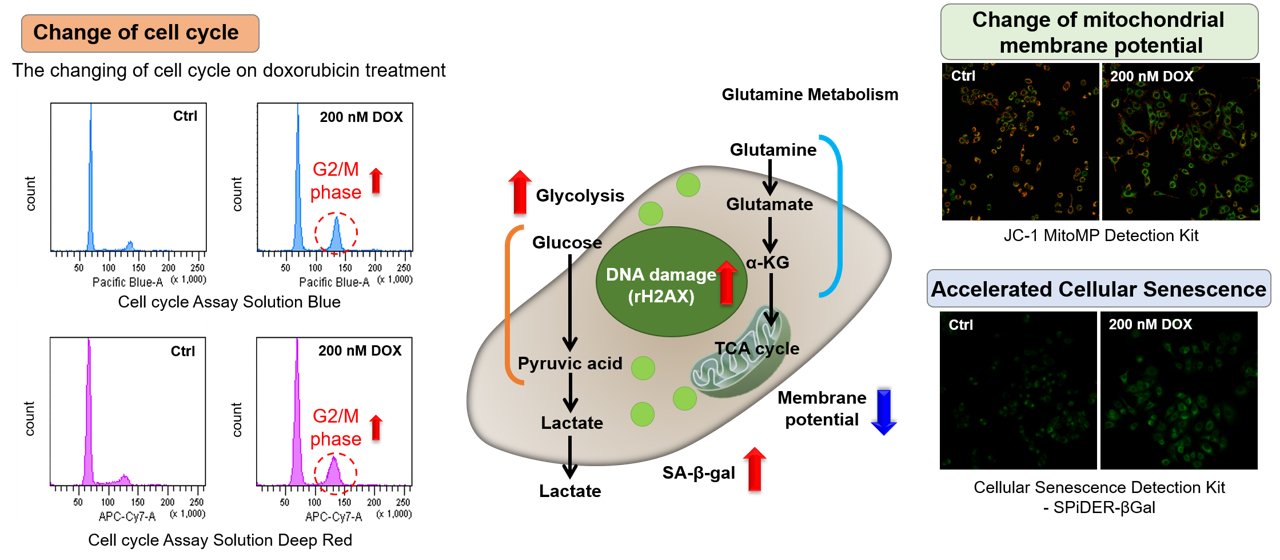
Association between cellular senescence and cell cycle
Doxorubicin (DOX) acts to inhibit cell proliferation during G2/M phases of the cell cycle and induces cellular senescence. After adding DOX to A549 cells, higher histogram peaks for the G2/M phase (Cell Cycle Assay Solution Blue and Deep Red), induces cellular senescence (Cellular Senescence Detection Kit - SPiDER-βGal), and the differences in mitochondrial membrane potential (JC-1 MitoMP Detection Kit) were observed.
Quantification with confocal quantitative image cytometer
In the conventional method of X-gal, SA-β-gal-positive cells are counted under microscope and calculate the percent of the senescent cells by compared with total cells. The SA-β-gal-positive cells were stained with this kit and analyzed using confocal quantitative image cytometer CQ1(Yokogawa Electric Corporation).
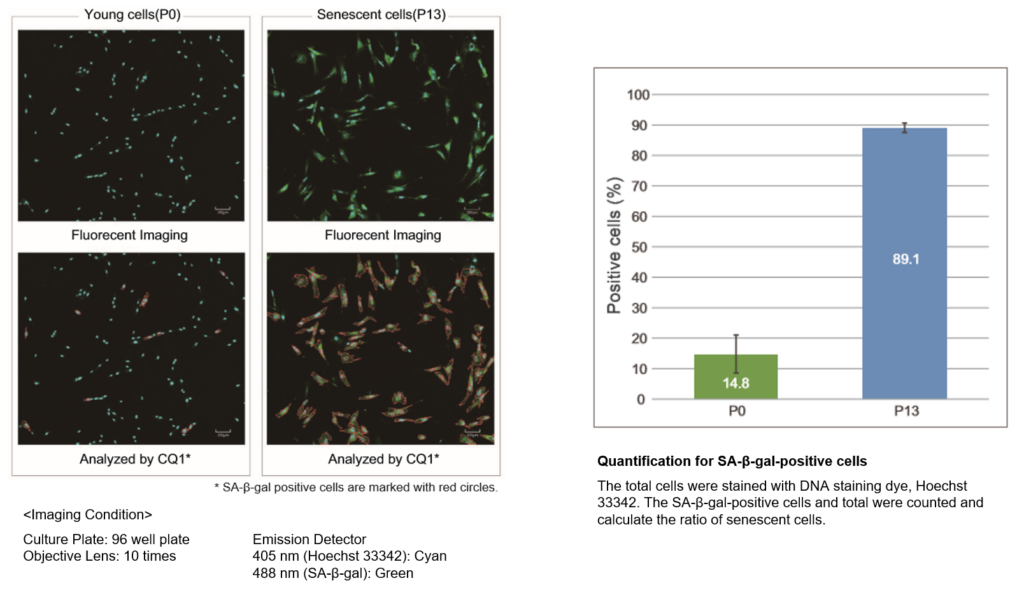
The difference of SA-β-gal-positive cells ratio were shown in WI-38 cells depending on the number of passage. The data was quickly analysed with the confocal quantitative image cytometer compared with the manually counting procedure with X-gal staining method.
参考文献
Instrument: Microscopy Flow Cytometry
Citation(Link): T. Doura, M. Kamiya, F. Obata, Y. Yamaguchi, T. Y. Hiyama, T. Matsuda, A. Fukamizu, M. Noda, M. Miura, Y. Urano, "Detection of LacZ-Positive Cells in Living Tissue with Single-Cell Resolution.", Angew Chem Int Ed Engl., 2016, 55, 33
2) Sample Type: Tissue (Mouse Adipose Tissue)
Instrument: Microscopy
Citation(Link): T. Sugizaki, S. Zhu, G. Guo, A. Matsumoto, J. Zhao, M. Endo, H. Horiguchi, J. Morinaga, Z. Tian, T. Kadomatsu, K. Miyata, H. Itoh & Y. Oike, "Treatment of diabetic mice with the SGLT2 inhibitor TA-1887 antagonizes diabetic cachexia and decreases mortality", Nature Partner Journal:Aging and Mechanisms of Disease., 2017, doi:10.1038/s41514-017-0012-0.
3) Sample Type: Cell (HSP27-knockdown) Protein (Ki67, cyclin B1)
Instrument: Microscopy
Citation(Link): A. Park, I. Tsunoda and O. Yoshie, "Heat shock protein 27 promotes cell cycle progression by down-regulating E2F transcription factor 4 and retinoblastoma family protein p130", J. Biol. Chem., 2018, doi: 10.1074/jbc.RA118.003310 .
4) Sample Type: Cell (A549)
Instrument: Microscopy Flow Cytometry
Citation(Link): R. Tanino, Y. Tsubata, N. Harashima, M. Harada and T. Isobe, "Novel drug-resistance mechanisms of pemetrexed-treated non-small cell lung cancer", Oncotarget., 2018, 9, (24), 16807.
5) Sample Type: Cell(NHDF)
Instrument: Microscopy
Citation(Link): Y. Kitahiro, A. Koike, A. Sonoki, M. Muto, K. Ozaki and M. Shibano. , "Anti-inflammatory activities of Ophiopogonis Radix on hydrogen peroxide-induced cellular senescence of normal human dermal fibroblasts.", J Nat Med., 2018, 72, 905.
6) Sample Type: Tissue(Aged Mouse Intestinal Epithelium Organoid)
Instrument: Microscopy
Citation(Link): R. Uchida, Y. Saito, K. Nogami, Y. Kajiyama, Y. Suzuki, Y. Kawase, T. Nakaoka, T. Muramatsu, M. Kimura and H. Saito, "Epigenetic silencing of Lgr5 induces senescence of intestinal epithelial organoids during the process of aging", NPJ Aging Mech Dis., 2018, doi:10.1038/s41514-018-0031-5.
7) Sample Type: Tissue(Frozen Kidney Slide)
Instrument: Microscopy
Citation(Link): S. R. Kim, A. Eirin, X. Zhang, A. Lerman and L. O. Lerman, "Mitochondrial Protection Partly Mitigates Kidney Cellular Senescence in Swine Atherosclerotic Renal Artery Stenosis.", Cell. Physiol. Biochem., 2019, 52, 617.
8) Sample Type: Cell(HN6, HN12, HN13)
Instrument: Flow Cytometry
Citation(Link): Liana P. Webber, Veronica Q. Yujra, Pablo A. Vargas, Manoela D. Martins. Cristiane H. Squarize, Rogerio M. Castilho, "Interference with the bromodomain epigenome readers drives p21 expression and tumor senescence", Cancer Letters., 2019, doi.org/10.1016/j.canlet.2019.06.019.
9) Sample Type: Cell(UE7T-13)
Instrument: Flow Cytometry
Citation(Link): H. Ise, K. Matsunaga, M. Shinohara and Y. Sakai, Improved Isolation of Mesenchymal Stem Cells Based on Interactions between N-Acetylglucosamine-Bearing Polymers and Cell-Surface Vimentin ", Stem Cells Int., 2019, 4341286, 13.
10) Sample Type: Cell(Mouse Corneal stroma)
Instrument: Flow Cytometry
Citation(Link): X. Wang, M. Qu, J. Li, P. Danielson, L. Yang and Q. Zhou, Induction of Fibroblast Senescence During Mouse Corneal Wound Healing.", Invest. Ophthalmol. Vis. Sci., 2019, 60, (10), 3669.
11) Sample Type: Cell(VZ/SVZ)
Instrument: Flow Cytometry
Citation(Link): Y. Nakatani, H. Kiyonari and T. Kondo, Ecrg4 deficiency extends the replicative capacity of neural stem cells in a Foxg1-dependent manner.", Development., 2019, 146, (4), 18.
12) Sample Type: Cell(HaCaT, HEK001)
Instrument: Microscopy
Citation(Link): Y. S. Ryu, K. A. Kang, M. J. Piao, M. J. Ahn, J. M. Yi, G. Bossis, Y. M. Hyun, C. O. Park and J. W. Hyun, Particulate matter-induced senescence of skin keratinocytes involves oxidative stress-dependent epigenetic modifications", Exp. Mol. Med., 2019, 51, 108.
13) Sample Type: Cell(HT1080)
Instrument: Microscopy
Citation(Link): E. M. Angela Ibler, E. Mohamed, L. N. Kathryn, A. B. Natalia, F. E. K. Sherif and H. Daniel, Typhoid toxin exhausts the RPA response to DNA replication stress driving senescence and Salmonella infection', Nat Commun., 2019, 10, 4040.
14) Sample Type: - (Review)
Instrument: Flow Cytometry
Citation(Link): B.L. Torres, A. Estepa-Fernandez, M. Rovira, M. Oraez, M. Serrano, R. Martinez-Manez and F. Sancenon,"The chemistry of senescence.", The chemistry of senescence., 2019, (3), 426-411
15) Sample Type: Cell(PC12)
Instrument: Microscopy
Citation(Link): N. Wang, H. Wang, L. Li, Y. Li and R. Zhang, "β-Asarone Inhibits Amyloid-β by Promoting Autophagy in a Cell Model of Alzheimer's Disease.", Front Pharmacol., 2020, 10, 1529
16) Sample Type: Cell(A2780)
Instrument: Flow Cytometry
Citation(Link): Z. Wang, J. Gao, Y. Ohno, H. Liu and C. Xu, "Rosiglitazone ameliorates senescence and promotes apoptosis in ovarian cancer induced by olaparib.", Cancer Chemother Pharmacol., 2020.
17) Sample Type: Tissue (Mouse Frozen Kidney Slide)
Instrument: Microscopy Flow Cytometry
Citation(Link): J. H. Cho, E. Kim, Y. Son, D. Lee, Y. S. Park, J. H. Choi, K. Cho, K. Kwon and J. Kim, "CD9 Induces Cellular Senescence and Aggravates Atherosclerotic Plaque Formation.", Cell Death Differ., 2020, doi: 10.1038/s41418-020-0537-9
18) Sample Type: Cell(T cell)
Instrument: Flow Cytometry
Citation(Link): S. Yoshida, H. Nakagami, H. Hayashi, Y. Ikeda, J. Sun, A. Tenma, H. Tomioka, T. Kaawano, M. Shimamura, R. Morishita and H. Rakugi, "The CD153 vaccine is a senotherapeutic option for preventing the accumulation of senescent T cells in mice.", Nat. Commun., 2020, 11, (2482), doi:10.1038/s41467-020-16347-w
19) Sample Type: Cell(PC12)
Instrument: Microscopy
Citation(Link): N. Wang, H. Wang, L. Li, Y. Li and R. Zhang, "β-Asarone Inhibits Amyloid-β by Promoting Autophagy in a Cell Model of Alzheimer's Disease.", Front Pharmacol., 2020, 10, 1529
20) Sample Type: Cell(ARPE-19)
Instrument: Microscopy
Citation(Link): T. Yamazaki, H. Suzuki, S. Yamada, K. Ohshio, M. Sugamata, T. Yamada and Y. Morita, "Lactobacillus paracasei KW3110 Suppresses Inflammatory Stress-Induced Premature Cellular Senescence of Human Retinal Pigment Epithelium Cells and Reduces Ocular Disorders in Healthy Humans”, Int J Mol Sci, 2020, 21(14), 5091