PHOSPHOLIPASE D [PLDP]
(Glycerophospholipid specific)
fromStreptomyces sp.
(Phosphatidylcholine phosphatidohydrolase, EC 3.1.4.4)

Preparation and Specification
- Appearance
- : Light brownish amorphous powder, lyophilized
- Specific activity
- : More than 100 U/mg solid
Properties
- Substrate specificity
- : See Table 1
- Molecular weight
- : 46 kDa (gel filtration)
- Isoelectric point
- : pH 4.2
- Optimum pH
- : 5.5–6.0Figure 1
- pH stability
- : 4.2–8.5 (37℃, 60 min, 0.05% BSA) Figure 2
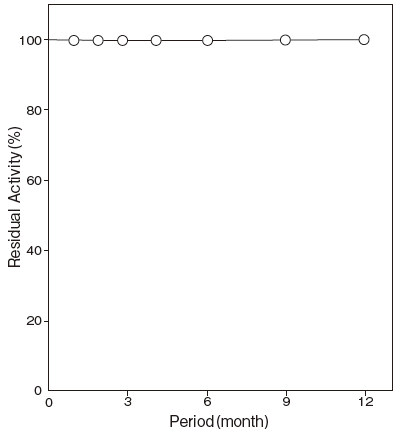
- Storage stability
- : At least one year at -20℃Figure 3
- Effect of various
chemicals - : See Table 2 and Table 3
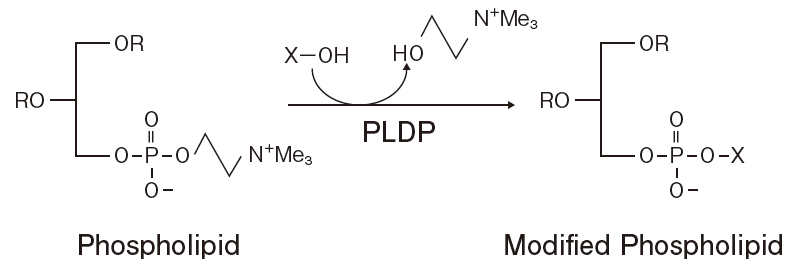
- Transphosphatidylation Catalyzed by Phospholipase DFigure 4
Figure 5
Figure 6
Table 1. Substrate specificity
| Substrate | Relative activity (%) |
|---|---|
| Lecithin | 100 |
| Lysolecithin | 3.4 |
| Sphingomyelin | 0.03 |
Table 2. Effect of detergents on PLDP activity
| Additive | Concentration (%) | Relative activity (%) |
|---|---|---|
| None | 0 | 0 |
| Triton X–100 | 0.1 | 28 |
| 0.5 | 100 | |
| 1.0 | 78 | |
| SDS | 0.1 | 15 |
| Deoxycholate | 0.1 | 8 |
Table 3. Effect of metal ions on PLDP activity
| Metal ion | Concentration (mM) | Relative activity (%) |
|---|---|---|
| None | 0 | 100 |
| CaCl2 | 10 | 99 |
| MgCl2 | 10 | 101 |
| MnCl2 | 10 | 132 |
| ZnCl2 | 1 | 99 |
| CoCl2 | 1 | 106 |
| BaCl2 | 1 | 101 |
| CuCl2 | 1 | 65 |
| EDTA | 1 | 99 |
| EDTA | 10 | 100 |
Fig.1 pH Optimum
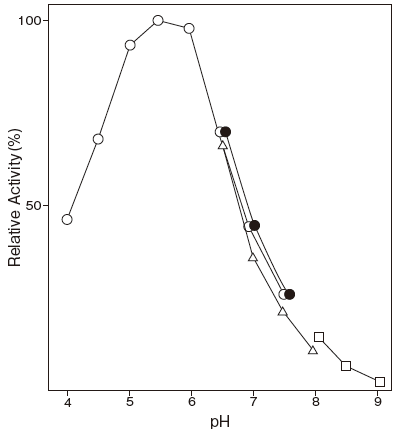
buffer
●: Phosphate buffer
△: Tris-HCI buffer
□: Glycine-NaOH buffer
Fig.2 pH Stability
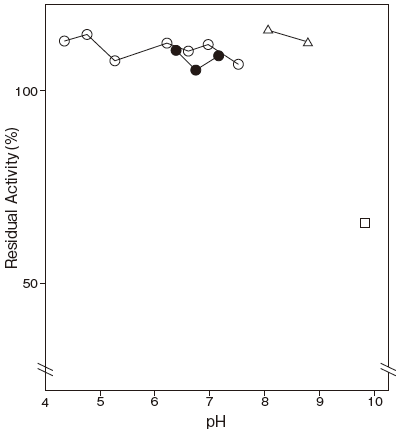
〇: 3,3-Dimethylglutarate-NaOH buffer
●: Phosphate buffer
△: Tris-HCI buffer
□: Glycine-NaOH buffer
Fig.3 Storage (lyophilized powder)
Transphosphatidylation Catalyzed by Phospholipase D
| Organic phase : | Solvent (0.5ml) Contained 1mg Dipalmitoylphosphatidylcholine |
| Aqueous phase : | 0.25M Cytidine–HCl buffer pH4.5 (0.1ml) and 0.05ml of water contained 0.9 Unit of PLDP |
| Reaction Temp : | 35℃, Reaction : 90 min. |
| Fig.4 | The Influence of Organic Solvents on the Yield of 5’-Phosphatidylcytidine by PLDP-Catalyzed Transphosphatidylation |
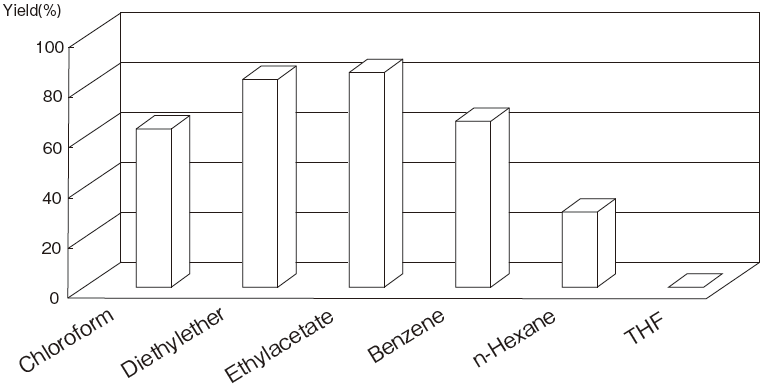
Fig.5 Optimum pH of PLDP-Catalyzed
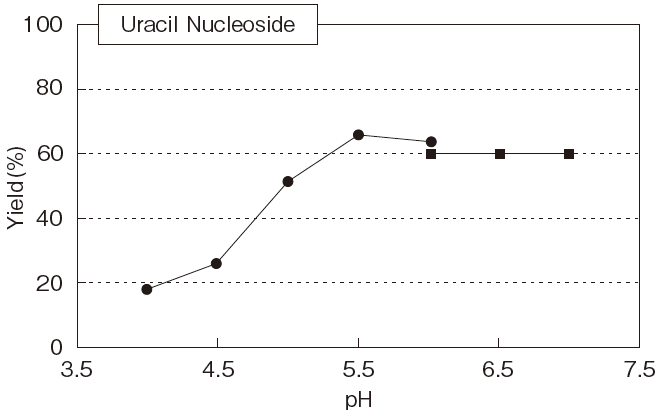
pH6-7 : MES-NaOH buffer
pH4-6 : Acetate buffer
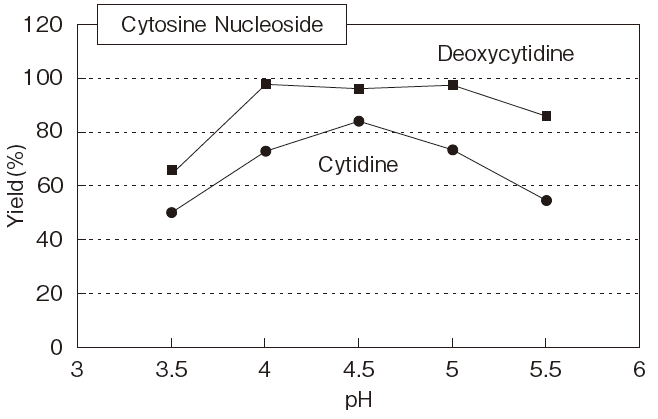
Cytidine, Deoxycytidine : 250mM
pH4-5.5 : Acetate buffer
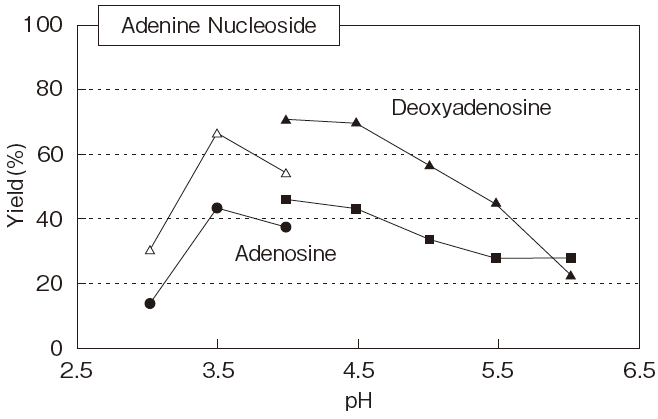
Adenosine : 50mM
pH4-6 : Acetate buffer
Deoxyadenisine : 50mM
| Phospholipid acceptor : | Dipalmitoylphosphatidylcholine in Chloroform (1ml) |
| Buffer : | 0.5ml contained 1.8 Units of PLDP |
| Reaction Temp : | 35℃ |
| Reaction period : | 90 min. |
| Fig.6 | The Influence of Reaction Temperature on the Yield of 5’-Phosphatidylcholine by PLDP-Catalyzed Transphosphatidylation (Donor:dipalmitoylphosphatidylcholine, Acceptor:Cytidine, pH4.5) |
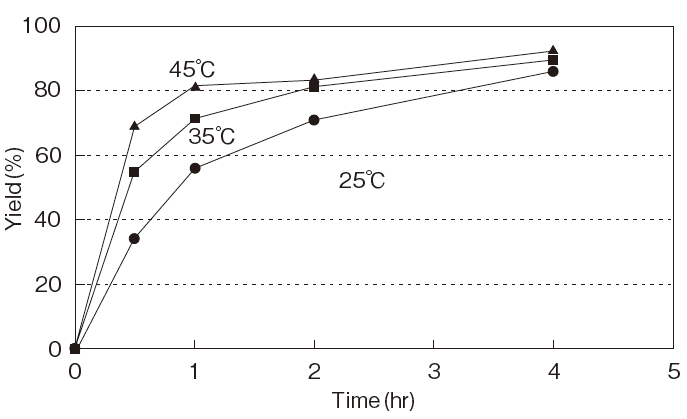
| Fig.7 | The Effect of Acceptor-Donor Ratio on the Yield of 5’-Posphatidylcytidine by PLDP-Catalyzed Transphosphatidylation |
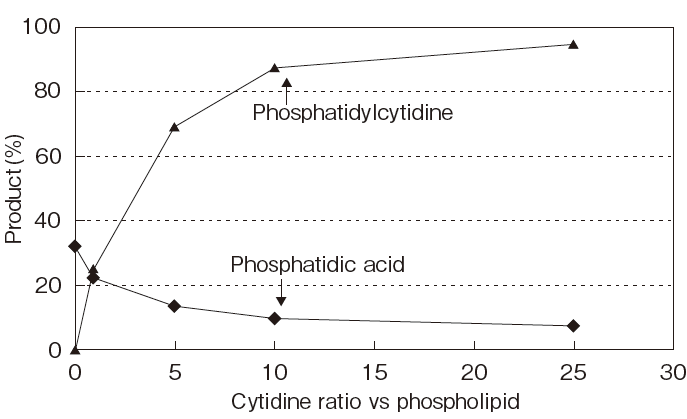
| Organic solvent : | 10 mM Dipalmitoylphosphatidylcholine in Chloroform (1ml) |
| Aqueous solvent : | 0.1 M Glycine–HCl (pH4.5) 0.5ml containing 1.8 Units of PLDP |
| Reaction : | at 35℃, with continuous stiiring, for 180min. |
Assay
Principle
The assay is based on the increase in absorbance at 500 nm as the formation of quinoneimine dye proceeds in the following reactions:
| PLDP | ||
| Phosphatidyl choline +H2O | → | Phosphatidic acid+Choline |
| COD | ||
| Choline +2 O2+H2O | → | Betaine+2H2O2 |
| POD | ||
| 2 H2O2+Phenol+4–AA | → | Quinoneimine dye+4 H2O |
COD: Choline oxidase
Unit definition
One unit is defined as the amount of enzyme which hydrolyzes 1 μmole of phosphatidylcholine to phosphatidic acid and choline per minute at 37℃ under the conditions specified in the assay procedure.
Reagents
- Reaction mixture for the first reaction
0.2 M DMG–NaOH buffer pH 5.5 0.10 ml 10 mM CaCl2 0.05 ml 3% (W/V) Triton X–100 solution 0.05 ml 10 mM Substrate solution 1) 0.10 ml Distilled water 0.15 ml DMG: 3, 3’–Dimethylglutarate 1) : 10 mM Substrate solution
Dissolve 78.6 mg of 1,2–Dioleoyl–sn–glycero–3–phosphocholine with 10 ml of 5% Triton X–100 (W/V) - Reaction mixture for the second reaction
15 mM 4–AA 0.05 ml 0.2% (W/V) Phenol 0.05 ml 1 M Tris–HCl buffer, pH 8.0 0.05 ml 50 U/ml POD 2) 0.05 ml 50 U/ml COD 3) 0.05 ml Distilled water 0.25 ml 2) : 50 U/ml POD
Dissolve 500 U (PPU) of POD with 10 ml of distilled water.3) : 50 U/ml COD
Dissolve 500 U of COD with 10 ml of 10 mM Tris–HCl buffer, pH 8.0. - Reaction stopper
1 M Tris–HCl buffer containing 10 mM EDTA and
1% (W/V) Cetyltrimetylammonium chloride
EDTA: Ethylenediamine tetraacetic acid - Reaction dilution solution
1% (W/V) Triton X–100 - Enzyme dilution buffer
10 mM DMG–NaOH buffer, pH 5.5 containing 0.1%Triton X–100 - Reagents:
DMG : Tokyo Kasei Kogyo Co., Ltd. #D1322
Triton X–100: The Dow Chemical Company
1,2–Dioleoyl–sn–glycero–3–phosphocholine:Sigma Chemical Co. #P–6354EDTA (2Na・2H2O) : KISHIDA CHEMICAL Co., Ltd.#060–29133COD: Nagase Diagnostics Co., Ltd. #T–05
4–AA: NACALAI TESQUE, INC. Special grade #01907–52
Cethyltrimethylammonium chloride:FUJIFILM Wako Pure Chemical CorporationPOD: Sigma Chemical Co. Type Ⅱ #P–8250
#087–06032
Enzyme solution
Weigh about 20 mg of test sample exactly and add enzyme dilution buffer to make a total of 20 ml. Dilute it with enzyme dilution buffer to adjust the concentration as required.
Procedure
- Pipette 0.45 ml of reaction mixture for the first reaction into a small test tube and preincubate them at 37℃.
- After 5 min, add exactly 50 μl of enzyme solution and mix to start the first reaction.
※ In the case of a test blank, add 50 μl of enzyme dilution buffer in place of enzyme solution at this point. - After 10 min, add 0.50 ml of reaction stopper and mix.
On stopping the first reaction, add 0.50 ml of the reaction mixture for the second reaction immediately to start the second reaction at 37℃. - After 20 min, add 1.5 ml of reaction dilution solution and mix.
- After 10 min, measure the absorbance at 500 nm.
△A = (As-Ab) ≦0.40 AbsAbsorbance sample : As blank : Ab
Calculation
- Activity (U/mg) = {(△A/10)/(12.2×1/2)} × 3.00/0.05 × 1/x
12.2 : millimolar extinction coefficient of quinoneimine dye at 500 nm (cm2/ μmole)2 : the multiplier derived from the fact that 1 mole of phosphatidyl choline produces 2 mole of H2O2 1/2 : the multiplier derived from the fact that 2 mole of H2O2 produces 1 mole of quinoneimine dye 10 : reaction time (min) 3.00 : final volume (ml) 0.05 : volume of enzyme solution (ml) X : concentration of the sample in enzyme solution ( mg/ml)
Storage
Storage at - 20℃ in the presence of a desiccant is recommended. Enzyme activity will be retained for at least one year under this condition (Figure 3) .
References
- Satosi S., Shigeyuki I., Hideo S., and Jun-ichi M., (1987)
Chem. Pharm. Bull., 35. 1, 447–449 - Satoshi S., Shigeru U., Shigeyuki I., Kiyofumi F., Akira M., and Tohru U., (1987) Tetrahedron, 28, 2, 199–202
- Satoshi S., Hiromichi I., Shigeru U., Shigeyuki I.,
Kiyofumi F., Masatoshi T., Akira M., and Tohru U.,
(1988) Chem. Pharm. Bull., 36, 1, 209–217 - Satoshi S., Shigeyuki I., Kiyofumi F., and Tohru U. (, 1988)
Chem. Pharm. Bull., 36, 12, 5020–5023 - Satoshi S., Hiromichi I., Atsushi S., Keishi N., Shigeyuki I., and Akira M., (1995) Bioorg. Med. Chem., 3, 3, 235–243
PLDP 活性測定法 (Japanese)
試薬液
- 第一反応試薬混合液
0.2M DMG–NaOH 緩衝液 pH5.5 0.10 ml 10mM 塩化カルシウム溶液 0.05 ml 3% (W/V) トリトンX–100 溶液 0.05 ml 10mM 基質溶液1) 0.10 ml 精製水 0.15 ml 1) : 10mM 基質溶液
1, 2 - ジオレオイルsn– グリセロ–3– ホスホコリン 78.6mg を5% (W/V) トリトンX–100 溶液10ml で溶解する。 - 第二反応試薬混合液
15mM 4–AA 溶液 0.05 ml 0.2% (W/V) フェノール液 0.05 ml 1M トリス–HCl 緩衝液 pH8.0 0.05 ml 50U/ml POD 溶液2) 0.05 ml 50U/ml COD 溶液3) 0.05 ml 精製水 0.25 ml 2) : 50U/ml POD 溶液
POD500 単位 (PPU) を精製水10ml で溶解する。3) : 50U/ml COD 溶液
COD500 単位 (U) を10mM トリス-HCl 緩衝液pH8.0 10ml で溶解する。 - 反応停止液
1% (W/V) 塩化セチルトリメチルアンモニウムと10mM EDTA を含む1M トリス–HCl 緩衝液pH8.0 - 反応用希釈液
1% (W/V) トリトンX–100 溶液 - 酵素溶解希釈用液
0.1% トリトン X–100 を含む10mM DMG–NaOH
緩衝液pH5.5 - 試薬
DMG (3,3– ジメチルグルタール酸) :東京化成製#D1322トリトンX–100:Dow Chemical 製
1,2– ジオレオイルsn– グリセロ–3– ホスホコリン:シグマ製 #P–6354EDTA (エチレンジアミン四酢酸・2Na・2H2O) :キシダ化学製 #060–29133COD (コリン酸化酵素) :ナガセダイアグノスティックス製 #T–05
4–AA:ナカライテスク製 特級 #01907–52
塩化セチルトリメチルアンモニウム:富士フイルム和光純薬製 #087–06032POD:シグマ製 Type Ⅱ #P–8250
酵素試料液
- 検品約20mg を精密に量り、酵素溶解希釈用液で全容20ml とする。
その液を酵素溶解希釈用液で適宜希釈する。
測定操作法
- 小試験管に第一反応試薬混合液0.45ml を正確に分注し、37℃で予備加温する。
- 5 分経過後、酵素試料液50 μl を正確に加えて混和し、37℃で第一反応を開始する。
※ 盲検はこの時点で酵素試料液の代わりに酵素溶解希釈用液50 μl を加える。 - 10 分経過後、反応停止液0.50ml を加えて混和し、第一反応を停止すると共に、直ちに第二反応試薬混合液0.50ml を加えて混和し、37℃で第二反応を開始する。
- 20 分経過後、反応用希釈液1.50ml を加えて混和する。
- 10 分経過後、500nm における吸光度を測定する。
求められた吸光度を試料液はAs、盲検液はAb とする。
ΔA = (As-Ab) ≦0.40 Abs
計算
活性 (U/mg) = {(ΔA/10)/(12.2×1/2)} × 1/2 × 3.00/0.05 × 1/x| 12.2 : | キノンイミン色素の500nm におけるミリモル分子吸光係数 (cm2 / μmole) |
| 2 : | フォスファチジルコリン1 モルからH2O2 2 モルが生成することによる係数 |
| 1/2 : | H2O2 2 モルからキノンイミン色素1 モルが生成することによる係数 |
| 10 : | 反応時間 (min) |
| 3.00 : | 反応総液量 (ml) |
| 0.05 : | 反応に供した酵素試料液量 (ml) |
| X : | 酵素試料液中の検品濃度 (mg/ml) |
